Recommendations Based on CAMBRELLA
The CAMbrella research team’s recommendations for health policy makers at both European and national levels are, again, in line with the WHO recommendations:
1) Perform high-quality research on the therapeutic effects of CAM, with an emphasis on the most widespread health problems of Europeans.
2) Quantify the possible economic benefits of using CAM within European healthcare.
3) Evaluate the possible models for integrating CAM into healthcare systems, including providing training and certifications.
4) Create a pan-European approach to CAM, including financial support for R&D within this sector.
(See page 99, more details on page 103 and the following pages.)
The proposal was substantiated by, among other things, the following arguments:
1) Due to the popularity of CAM in the EU, it is necessary to obtain and provide patients with reliable information on the effectiveness and safety of CAM methods and the quality of their providers. It is also necessary to ensure predictable conditions for the use and provision of CAM for patients and practitioners moving across EU countries.
2) European healthcare systems face a crisis linked to the demographic developments in Europe – an ageing population, chronic diseases and cancer. CAM can potentially provide solutions to preventing, treating and rehabiliting these diseases.
3) In comparison to other countries, CAM research and the potential health benefits for European citizens are underestimated and underfunded in Europe. (See page 103 and the following pages.)
“Recommendations for Policy Makers – CAMbrella calls on
the Members of the European Parliament, the European Commission, and the national health and research policy makers:
-
establish a European–wide approach to assess the prevalence of use of core CAM treatment disciplines
-
identify the citizens’ access to and preferences for CAM provision as well as their perspectives on education, training and practice of CAM providers
-
identify the most promising CAM treatment options for the most prevalent health conditions in Europe (chronic diseases like cancer, diabetes, musculoskeletal problems, obesity, and many others in an ageing population) with a clear emphasis on concurrent evaluation of CAM as an additional or alternative treatment strategy in real–world settings
-
quantify the economic effects of CAM in European healthcare
-
give clear guidance on CAM safety issues
-
research and evaluate different models of CAM healthcare integration into routine care programmes
-
address the diversity of training, education, regulation and provision of CAM across Europe
-
collect and disseminate valuable CAM research findings for the European citizens and CAM providers and the scientific community
-
foster the pan–European collaboration between CAM researchers by financially support of academic exchange and improve European CAM research capability by establishing career opportunities for excellent researchers in the field of CAM.” (61)
Based on the CAMbrella project, a CAMbrella output document was approved on 26 September, 2012 – A research roadmap for complementary and alternative medicine what we need to know by 2020 (62). The document is available in full on page 123 (and the following pages). Its most important suggestions are summarized below:
“Our vision for 2020 is that there is an evidence base that enables European citizens to make informed decisions about CAM, both positive and negative. This roadmap proposes a strategic research agenda for the field of CAM designed to address future European health care challenges.” (63)
Six basic research areas have been proposed (64):
1. Research into the prevalence of CAM in Europe (i.e., how Europeans use CAM).
2. Research into differences regarding citizens’ attitudes and needs towards CAM.
3. Research into safety of CAM.
4. Research into the comparative effectiveness of CAM (i.e., determine when CAM is a sensible alternative).
5. Research into effects of context and meaning.
6. Research into different models of CAM health care integration.
Other suggestions:
“We propose that the EU actively supports an EU-wide strategic approach that facilitates the development of CAM research. This could be achieved in the first instance through funding a European CAM coordinating research office…” (65)
“With the aim to develop sustainability as second step, a European Centre for CAM should be established that takes over the monitoring and further development of a coordinated research strategy for CAM…” (66)
It is necessary to emphasize that the document A research roadmap for complementary and alternative medicine what we need to know by 2020 is only a strategy recommended by the CAMbrella research team to the European Union. The European Commission has not yet issued a binding resolution based on the conclusions from this research. However, meetings are currently being held with both medical and non-medical CAM associations, in the European Commission and the European Parliament. The subject matter is being discussed with increasing frequency. Reports from some of the meetings are included in the following parts of the publication (see page 126 and the following pages).
The pan-European research network CAMBRELLA continues to promote this idea throughout the EU. It mediates communication between involved parties (patients, CAM providers, representatives of conventional medicine, funding institutions, etc.) and presents its findings at conferences and in the European media.
George Lewith is one of those who actively presents the CAMbrella project’s findings and the research team's recommendations to the public. George Lewith is a professor of medical research at Southampton University, England and one of the researchers involved in the CAMbrella project.
Here are some excerpts from his article Why we need to research the use of complementary medicine (67) published in one of Britain's most prestigious periodicals – The Guardian – in 2012. In his article, professor Lewith highlights the urgent need for a strategic pan-European approach to CAM in order to ensure its quality and its equal accessibility to all European citizens. However, it seems that there are still enormous differences between EU states regarding their approaches to CAM:
“The UK has one of the most organised approaches to registration and regulation for non-medically qualified practitioners who provide most of these clinical interventions. In the rest of the EU, particularly in Germany and France, these treatments are almost always part of medical practice in both the community and in hospitals.” (68)
“The lack of good quality research, indeed its total absence in some of the new EU members in eastern Europe countries, is a major problem.” (69)
At the same time, Lewith draws attention to the fact that, in comparison to other countries, this sector suffers from a lack of financing throughout the EU:
“While the United States, China, Australia and India have a clear, government-funded and strategic approach to CAM research, this is not the case within the EU. CAM is a neglected area of research with little academic support and it needs active encouragement.” (70)
“In order to achieve these objectives, an EU research office for CAM should be established. We need to grasp every opportunity to improve and develop our healthcare resources, while supporting and improving approaches to self-care especially for chronic long-term problems that can consume vast resources in ageing populations.” (71)
On March 5, 2013, prof. George Lewith introduced the CAMbrella project at a conference in the UK House of Commons organized by the UK Parliamentary Group on Integrated Healthcare (see p. 122).
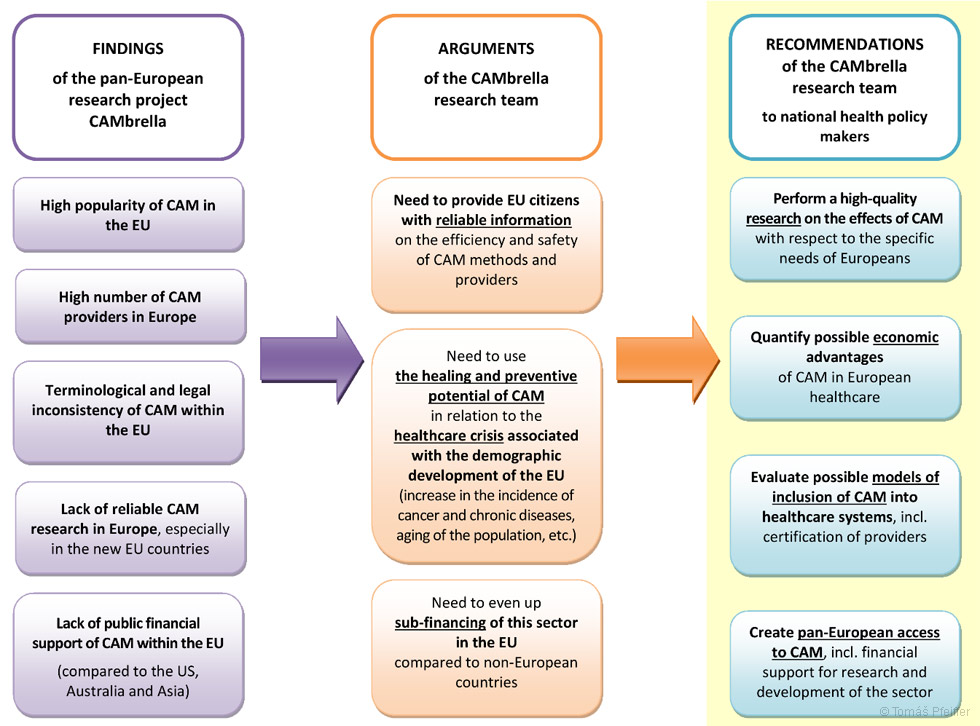
Figure 14 – Output of the pan-European CAMbrella project: findings on the CAM situation in the EU, recommendations for health policy makers (both at the national and European level) and the most important arguments. Author of the graphic and translation – KoS.



